
Octahedral Geometry: Six Electron Domains Connect the set of three with the set of two balloons by twisting at the points of connection for the two sets.Tie this remaining balloon to one of the balloon pairs.Tie balloons together to form two sets of two balloons.Trigonal Bipyramidal Geometry: Five Electron Domains Then, connect the two balloon pairs by twisting one pair’s connection around the other’s connection.Tetrahedral Geometry: Four Electron Domains Trigonal Planar Geometry: Three Electron Domains Inflate another balloon to the same size and tie it.Procedure Linear Geometry: Two Electron Domains Each atom connects to others using the appropriate molecular geometry. These products have colored atoms and use connectors to represent electron domains. Consider incorporating subsequent activities that use plastic molecular models.
Address the electron domain misconception as needed. Note: Students may incorrectly believe that the balloons in this activity represent electron clouds around the nucleus of an atom (rather than electron domains around a centrally-bonded atom). You can perform this activity as a demonstration, or alternatively, as a student investigation. When connected, balloons naturally adopt the lowest energy arrangement predicted by the VSEPR model.

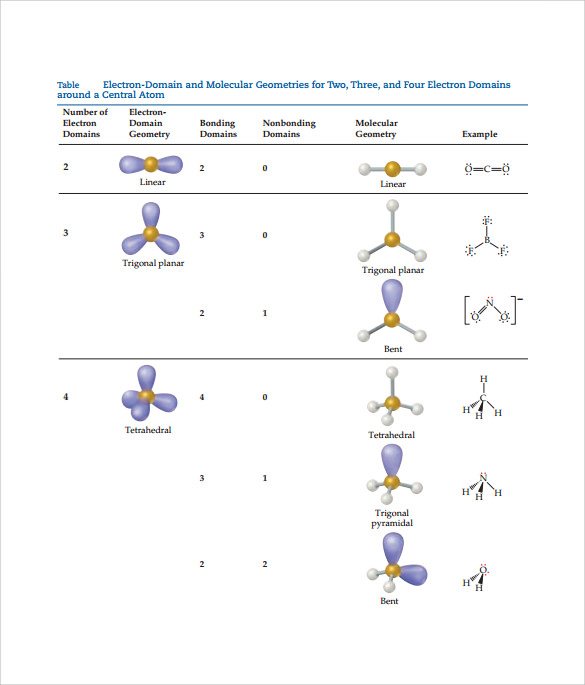
In this activity, each balloon represents an electron domain (either a bonding pair or a lone pair of electrons) surrounding a central atom. Three-dimensional models, however, give students the opportunity to view and manipulate models as needed, making molecular geometry much easier to understand. Two-dimensional representations of molecular geometries predicted by the valence-shell electron-pair repulsion (VSEPR) model can be difficult to grasp. Molecular geometry can be tricky for students to understand.
